BioKeralty news
NanoGSkin reduces grafting time thanks to a regenerative approach
The project points out that the clinical application of generated artificial skin would improve significantly patients’ survival and life quality
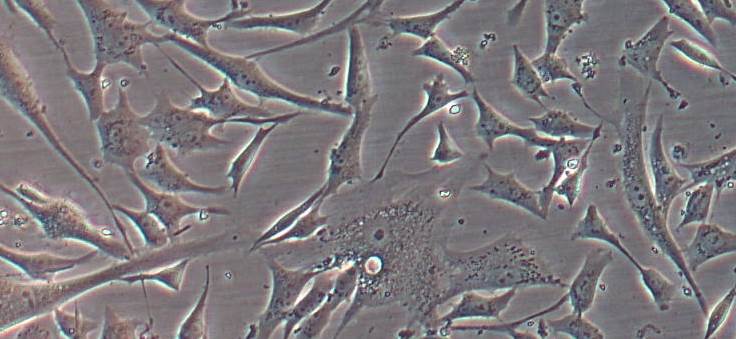
The image shows the absence of dermal histopathological conditions.
December 15 2021
The treatment of patients with serious burns affecting large skin areas becomes very complex and may even put in risk patient’s survival. However, after three years work and from regenerative and nanomedical approach, the NanoGSkin project has reduced grafting time and prevented wound infection. This result has been possible thanks to the joint work of a multidisciplinary consortium formed by the Technological University of Compiegne, the University of Bordeaux, the Biosanitary Research Institute of Granada, the Istituto Biochimico Italiano, the University of Galway, the University of Bordeaux and Keralty Health.
The project seeks to promote cell multiplication
to increase the effectiveness and tolerance of autograft
According to some first results, the clinical application of the artificial skin could improve significantly patients’ survival and life quality, offering regenerative hope to patients with serious burns. At the same time, from a technical point of view, it was possible to reduce the time of cell production adding nano-encapsulated growth factors, fact that also helps to grafting time.
In summary, NanoGSkin project’s main milestones were the following ones:
1. Manufactured production and expansion process of Nanoparticles (NP): a batch of rhEGF nanoparticles were successfully released under similar conditions of GMP, fulfilling in this way the specifications and requirements of GMP regulations. In addition, the manufacture of LNP-antibiotics using microfluidic equipment has proven to be an automated method, easy to control and replicate, offering at the same time the possibility of scaling for the pharmaceutical industry.
2. Confirmation of the bioengineered human skin substitute’s biosafety combined with nanoparticles: the project showed that the addition of nanoparticles to human skin substitutes does not affect the structure and functionality of the artificial tissue. Nanoparticles’ biosafety in vivo studies have confirmed the absence of histopathological dermic conditions.
3. Confirmation of the efficacy of bioengineered human skin surrogate in combination with NPs: efficacy in vitro studies with rhEGF-NPs and antibiotic-NPs have shown that charged nanoparticles can be used to cause the proliferation of human skin keratinocytes and obtain antibacterial beneficial effects.
4. Business plan: the viability of the products and the market acceptance of the functional substitute for skin have been also analyzed. In this sense, a business and commercialization plan were prepared, including as well a preliminary cost study.
Due to the combination of cell therapy and nanotechnology, both areas of interest for many clinicians and patients, Keralty group considers particularly promising the development of such an innovative proposal. As a result, it is expected that, in the near future, this therapy will be used with those patients who present specially a serious case in order to take benefit from the latest advances related to artificial skin. This is the reason why BioKeralty, one of the group’s R&D units, has been in charge of the development of antibiotics charged nanoparticles and growth factors, later integrated into the model, as well as the design and execution of toxicology tasks.